The presence of mutations in the TP53 gene is a powerful prognostic and predictive marker in chronic lymphocytic leukemia (CLL). Widespread use of NGS has enabled the detection of variants ≤10 % variant allelic frequency (low-VAF variants); however, the overall reliability and reproducibility of NGS techniques to identify such variants have been questioned repeatedly. Individual studies using sensitive, custom NGS-based assays have mostly demonstrated the shortened overall survival (OS) and event-free survival in patients with low-VAF TP53 variants treated with chemoimmunotherapy (CIT) regimens with median survival ranging between that of TP53 variants >10 % VAF (high-VAF) and wild-type TP53 (wt- TP53).
Within an ERIC multicenter study, we tested the ability of NGS methods used in diagnostic and research laboratories to detect low-VAF TP53 variants and analyzed the impact of the identified low-VAF variants on patients' survival.
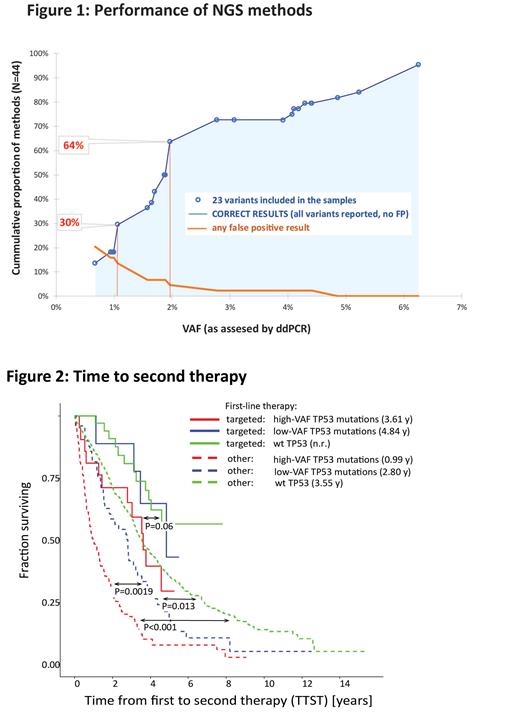
In the first phase of the study (Fig. 1), seven sample mixes containing 23 pathogenic TP53 variants (range, 0.7-6.3% VAF) were analyzed in 41 ERIC centers using 44 NGS-based assays. All variants were validated with droplet digital PCR (ddPCR); obtained values were used as a reference for the assessment of each NGS method's performance. NGS results were categorized as true positive (TP), false positive (FP; not present in original samples and reported by one center each), and not reported/false negative (FN). In total, laboratories reported 77.8% of all variants (784 out of 1008), reaching a sensitivity [TP/ (TP + FN)] of 85.6%, 94.5%, and 94.8% at 1%, 2%, and 3% VAF cut-off, respectively. While the VAFs of individual variants reported by laboratories varied, median values strongly correlated with ddPCR (R 2=0.9841). Thirty-eight FP variants were reported by 10 laboratories, mainly <2% VAF (23 FP of VAF ≤1%, 14 FP of VAF >1 and ≤2%, 1 FP > 2%). Individual feedback was provided to improve the methods' performance and to help set an appropriate detection limit.
In the second phase of the study, 12 centers provided results of TP53 NGS-based analysis of 1092 CLL clinical samples taken before first-line treatment (median time from sample to treatment 40 days). The impact of low-VAF variants (1-10% VAF; N=59) on time to second treatment (TTST; event: second treatment, death) and OS calculated from 1 st treatment initiation was compared to that of high-VAF variants (N=123) and wt- TP53 using logrank test with Benjamini-Hochberg correction of p-values. TTST (Fig. 2) of the low-VAF group was significantly shorter compared to wt- TP53 (P=0.013; median TTST wt- TP53 3.6 y, low-VAF 2.8 y, high-VAF 1 y) in patients not treated with targeted agents (N=999). If del(17p) status was considered, median TTST was the shortest in patients with a combination of del(17p) and either high (0.8 y) or low-VAF (1 y) TP53 mutations, followed by high-VAF (1.5 y) and low-VAF (2.8 y) mutations in the absence of del(17p) (P<0.001, P=0.032, P<0.001, P=0.026, respectively, compared to wt- TP53/no del(17p) (3.6 y)). In patients receiving frontline targeted agents (N=73; enriched for TP53 mutations), the results suggested shorter TTST for the high-VAF group only, but the difference was not significant (Fig. 2; P=0.06; median wt- TP53 n.r., low-VAF 4.8 y and high-VAF 3.6 y).
OS of patients with low-VAF variants was significantly shorter compared to the wt- TP53 group in patients never treated with targeted treatment (P=0.033; median OS wt- TP53 6.6 y, low-VAF 3.2 y and high-VAF 2.1 y). Targeted therapy in 2 nd or later therapy lines diminished the difference and only OS of the high-VAF group differed significantly from wt- TP53 (P<0.001; median OS wt- TP53 10.6 y, low-VAF 8.6 y, and high-VAF 5.1 y).
Altogether, we show that the cumulative reliability (no FN and FP) of methods tested increased continuously with VAF (Fig. 1), reaching 30% and 64% for variants ≥1.1% and 2% VAF, respectively. The reliability was affected by the type of NGS method and bioinformatic pipeline settings. We conclude that no strict threshold can be suggested from a technical standpoint. However, our results emphasize a strong need to validate/verify the NGS method, describe its limits, and report only reliable results. From a clinical standpoint, while low-VAF variants impact clinical outcomes for patients receiving CIT in the frontline setting, their clinical impact for patients treated with novel therapies remains to be evaluated in larger cohorts.
Disclosures
Brieghel:Octapharma: Other: Travel grant. Andres:AstraZeneca, Novartis, Roche, Janssen-Ciliag: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel support. Bellosillo:ThermoFisher: Research Funding, Speakers Bureau. Stilgenbauer:Amgen: Consultancy, Honoraria, Other: travel support, Research Funding; Abbvie: Consultancy, Honoraria, Other: travel support, Research Funding; Janssen: Consultancy, Honoraria, Other: travel support, Research Funding; Roche: Consultancy, Honoraria, Other: travel support, Research Funding; GSK: Consultancy, Honoraria, Other: travel support, Research Funding; Gilead: Consultancy, Honoraria, Other: travel support, Research Funding; Celgene: Consultancy, Honoraria, Other: travel support, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: travel support, Research Funding; Novartis: Consultancy, Honoraria, Other: travel support, Research Funding; Sunesis: Consultancy, Honoraria, Other: travel support, Research Funding. Tausch:Janssen-Cilag: Consultancy, Honoraria, Other: travel support, Speakers Bureau; AstraZeneca: Consultancy, Honoraria, Other: travel support, Speakers Bureau; BeiGene: Consultancy, Other: Travel support, Speakers Bureau; Roche: Consultancy, Honoraria, Research Funding, Speakers Bureau; Abbvie: Consultancy, Honoraria, Other: Travel Support, Research Funding, Speakers Bureau. Czekalska:Astra Zeneca: Honoraria. Chatzidimitriou:Novartis: Other; Jansenn: Other. Walewska:AbbVie, AstraZeneca, Janssen, Beigene: Other: meeting attendancies. da Silva:AstraZeneca: Research Funding; Janssen Cilag: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Takeda: Consultancy, Research Funding. Rossi:AbbVie, AstraZeneca, Gilead, BeiGene, BMS, Janssen, Lilly, Kyte: Honoraria, Research Funding. Baliakas:Gilead: Honoraria. Kahre:AstraZeneca Estonia: Honoraria. Alcoceba:Janssen, AstraZeneca: Honoraria, Other: Travel expenses. Scarfo:Octapharma: Speakers Bureau; Lilly: Consultancy; Janssen: Consultancy; BeiGene: Consultancy; AstraZeneca: Consultancy; AbbVie: Consultancy. Costa:Roche: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Astrazeneca: Consultancy, Honoraria; Genmab: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Janssen: Consultancy, Honoraria. Davi:Janssen, AstraZeneca: Honoraria. Tazon:Bristol Myer Squibb: Honoraria. Niemann:Carsten Niemann has received research funding and/or consultancy fees from AstraZeneca, Janssen, AbbVie, Beigene, Genmab, CSL Behring, Octapharma, Takeda, and Novo Nordisk Foundation.: Consultancy, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal